- Home
- 2016(9)
2016(9)
 |
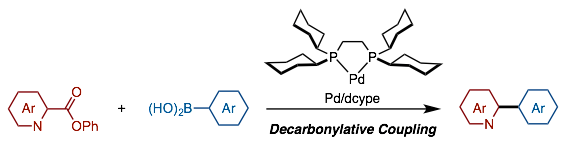 |
Muto, K.; Hatakeyama, T.; Itami, K.; Yamaguchi, J.
Org. Lett. 2016, 18, 5106–5109.
DOI 10.1021/acs.orglett.6b02556
Detail Palladium-Catalyzed Decarbonylative Cross-Coupling
 |
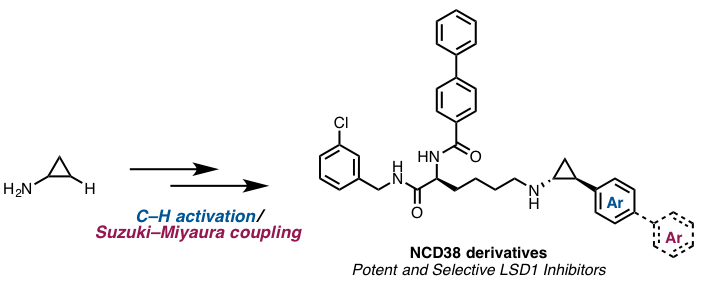 |
Miyamura, S.; Araki, M;; Ota, Y; Itoh, Y; Yasuda, S; Masada, M; Taniguchi, T; Sowa, Y; Sakai, T; Suzuki, T; Itami, K.; Yamaguchi, J.
Org. Biomol. Chem. 2016, 14, 8576–8585.
DOI 10.1039/C6OB01483F
Detail C-H Activation Enables Rapid SAR for LSD1 Inhibitors
 |
 |
Takise, R.; Itami, K.; Yamaguchi J.
Org. Lett. 2016, 18, 4428–4431.
DOI 10.1021/acs.orglett.6b02265
Detail Cyanization of phenol derivatives with metal-free cyanating agents
 |
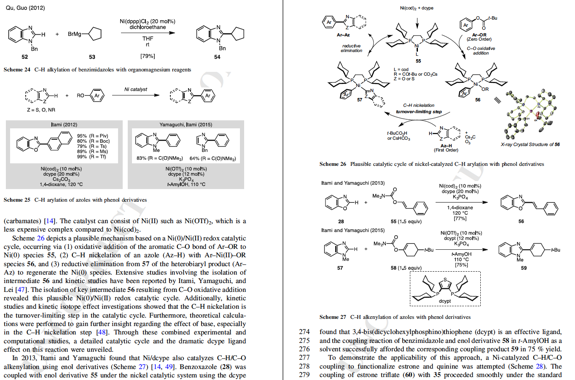 |
Yamaguchi, J*; Muto, K.; Itami, K.
Top Curr. Chem, 2016, 374, 55.
DOI 10.1007/s41061-016-0053-z
Detail Nickel-Catalyzed Aromatic C–H Functionalization
 |
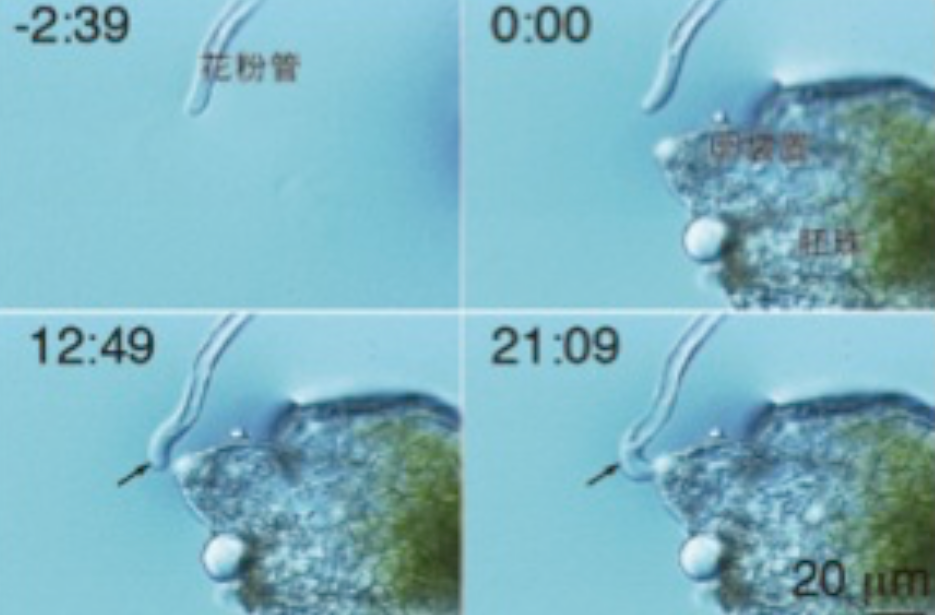 |
Mizukami, A. G.; Inatsugi, R.; Jiao, J.; Kotake, T.l Kuwata, K.; Ootani, K.; Okuda, S.; Sankaranarayanan, S.; Sato, Y.; Maruyama, D.; Iwai, H.; Garénaux, E.; Sato, C.; Kitajima, K.; Tsumuraya, Y.; Mori, H.; Yamaguchi, J.; Itami, K.; Sasaki, N.; Higashiyama, T.
10.1016/j.cub.2016.02.040
 |
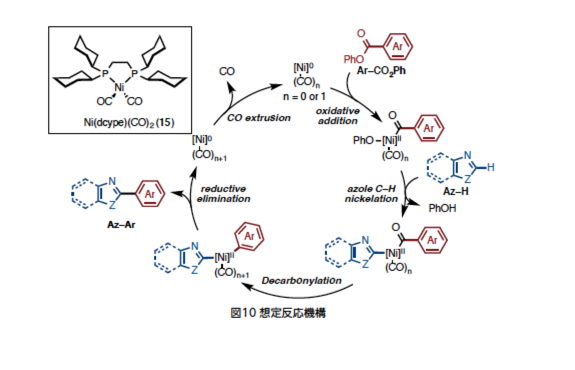 |
Yamaguchi, J.*; Muto, K.; Itami, K.
Chemical Times, 2016, 1–7.
Link Link
Detail Development and Elucidation of the Ni-Catalyzed Direct Coupling Reaction
 |
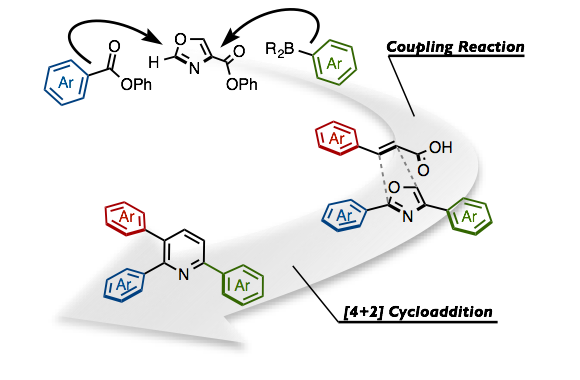 |
Amaike, K.; Itami, K.; Yamaguchi, J.
Detail Making Triarylpyridines
 |
 |
Yamaguchi, J.; Amaike, K.; Itami, K.
“Transition Metal-Catalyzed Heterocycle Synthesis via C-H Activation”、2016, Wiley-VCH
DOI 10.1002/9783527691920.ch16
Detail Synthesis of Natural Products and Pharmaceuticals via Catalytic C–H Functionalization


 日本語
日本語 中文
中文