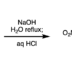Ring-Opening Fluorination of Bicyclic Azaarenes
Today:1views / Total:2,429views
Komatsuda, M.; Suto, A.; Kondo Jr., H.; Takada, H.; Kato, K.; Saito, B.; Yamaguchi, J.
Chem.Sci. 2022, Just Accepted.
DOI: 10.1039/d1sc06273e
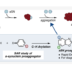
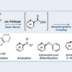
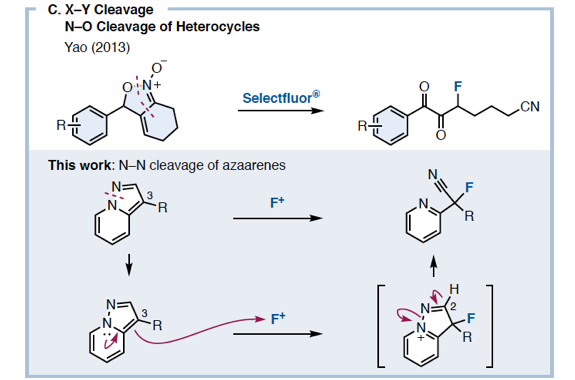
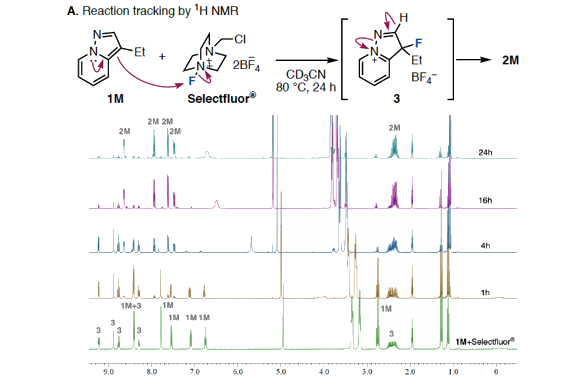
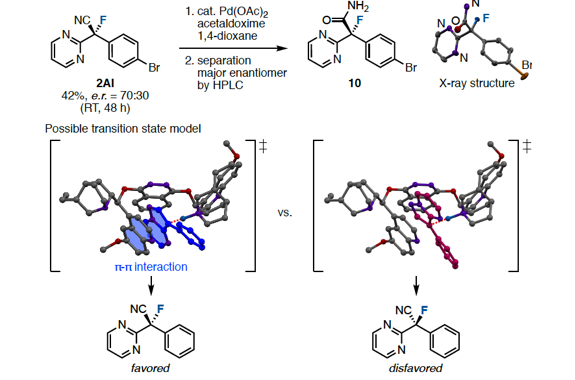
We have discovered a ring-opening fluorination of bicyclic azaarenes. Upon treatment of bicyclic azaarenes such as pyrazolopyridines with electrophilic fluorinating agents, fluorination of the aromatic ring is followed by a ring-opening reaction. Although this overall transformation can be classified as an electrophilic fluorination of an aromatic ring, it is a novel type of fluorination that results in construction of tertiary carbon–fluorine bonds. The present protocol can be applied to a range of bicyclic azaarenes, tolerating azines and a variety of functional groups. Additionally, mechanistic studies and enantioselective fluorination have been examined.



 日本語
日本語 中文
中文